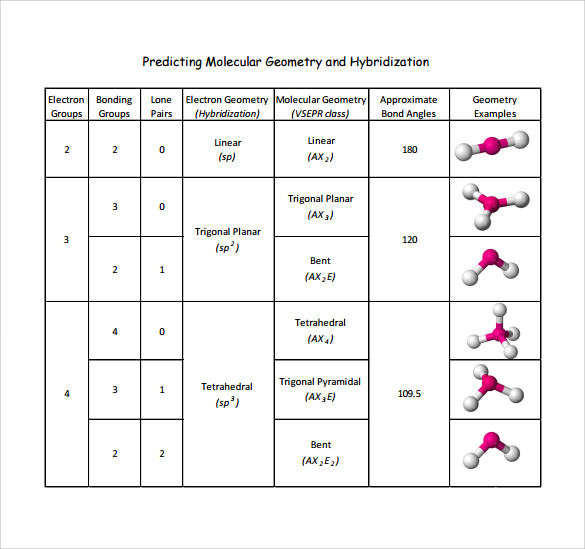
 Stephen McNeil at UBC Okanagan.Īll educational works available on this page are licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 2.5 Canada License. Whilst the atoms in carbon dioxide are held in a straight line, water is a bent molecule. However, their molecules are very different in shape. The similarities go even further - they are both partially formed from oxygen and they both contain covalent bonds. This page is maintained and copyright by W. Water and carbon dioxide are both triatomic molecules. Basic ArrangementĪnother VSEPR page, including a number of practice problems, Note that multiple bonds are counted as one region of electron density.ģ)ĝetermine the basic arrangement of all electron density regions (bonds + lone pairs) about the central atom.Ĥ) Determine the molecular geometry based on resulting positions of atoms. The resulting molecular geometry depends only upon the positions of the atoms, and will differ from the electron pair arrangement if there are lone pairs on the central atom.Ģ)Ĝount the bonds and lone pairs about the central atom. A common notation is used to classify various molecules and the shapes they should adopt: AX nE m, where A = central atom, X = atoms connected to A by bonds, E = lone pairs on A. The shape of the molecule can differ from the basic arrangement of electron density regions, depending on how many "corners" of the arrangement are taken up by lone pairs. There is a sharp distinction between ionic and covalent bonds when the geometric arrangements of atoms in compounds are. Most often Lewis structures of H2O are drawn with the two lone pairs angled above the oxygen, so this might be a more helpful way of drawing the structure so you remember that the shape is bent. The basic arrangements of the electron pairs depends on the number of regions of electron density (bonds + lone pairs), as follows: Regions of The molecular shape for H2O is bent because the lone pairs will repel each other more than they repel the bonded pairs. A simple biomembrane like erythrocyte contains well over hundred lipid species with diverse molecular shapes differing in the number of acyl chains.
0 Comments
Leave a Reply. |


 RSS Feed
RSS Feed
